Tetrahydrocannabivarin, or simply THCV, is a derivative of the cannabinoid known as Tetrahydrocannabinol (THC). While somewhat similar in chemical structure, they do have some distinct differences worth noting.
In today’s cannabinoid journal entry, we discuss Delta 9 Tetrahydrocannabivarin.
What is Tetrahydrocannabivarin?
Delta-9-tetrahydrocannabivarin (THCV) is a phytocannabinoid found in the cannabis plant. Discovered in minute quantities compared to other cannabinoids, THCV is a byproduct of the continuous breakdown of cannabigerolic acid (CBGA).
This compound has come under the auspices of researchers because of its multiple application and ability to perform the same functions of THC even better. At the same time, scientific research has identified novel components of compounds obtained from cannabinoids and their receptors for neuroprotection and regulation of neuronal function.
Related article: What is Delta 9?
THCV was discovered decades ago alongside other cannabidiols. In 1940, British scientist Robert Cahn discovered THC partially, and in 1986, R.K. Razdan took the research further by studying the structure-activity relationship (SAR) of the cannabis compounds.
We don’t who exactly discovered the THCV compound, but we know that its process of formation stems from CBGV.
During its final conversion, THCVA loses the molecular acid portion after the decarboxylation operation. The method of heat application over time is decarboxylation, also known as decarbonation. This process can occur either during the extraction, incineration, or vaporization process.
The molecule becomes THCV when subjected to light, heat, or time. A propyl (3 carbon) group exists in place of a pentyl (5 carbon) molecule group and breeds the difference between THCV and the THC. Although this difference can seem minor, THCV does not yield the same result as THC.
These effects include the reduction of anxiety attacks, suppression of hunger, and enhancement of bone growth.
The medicinal future of THCV is an optimistic one. Its ability to function better than the current foremost used cannabinoid, THC, is an advantage for the compound.
We need more research and clinical trials to give specifics on the compound and its use. THCV is one of four cannabinoids with identified potential as an anticonvulsant agent. Researchers have promised to look into this compound and gain more details in the nearest future.
How Does THCV Work in Our Bodies?
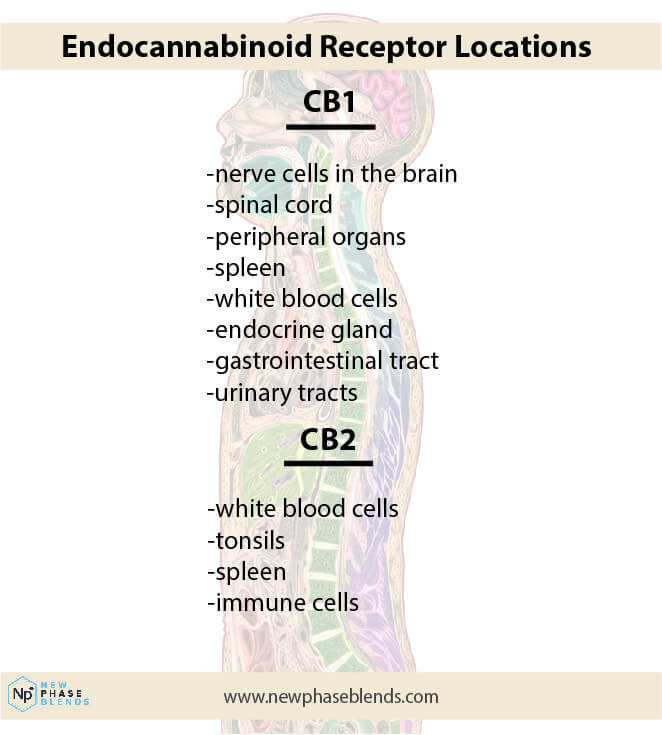
THCV functions as a receptor antagonist at CB1 and a partial receptor agonist at CB2.
THCV interacts with non-cannabinoid receptors 5HT1A in addition to its interactions with endocannabinoid receptors.
Related: How Does CBD Work in Our Bodies?
This stems from its antipsychotic influence. In addition, THCV bonds with multiple transient receptors that are possible pathways, including TRPV2, that can contribute to CBD and the cannabis plant extracts’ analgesic, anti-inflammatory, and anti-cancer impacts.
It has been found to have anti-epileptiform and anticonvulsant effects, which indicate potential use in the medical field to treat pathophysiological disorders such as uncontrolled epilepsy.
Possible Benefits of the Tetrahydrocannabivarin Cannabinoid
The profile of THCV is quite promising. Acute cannabinoid administration strengthened the disease’s effects in treating Parkinson’s disease in animals, including eliminating motor inhibitions.
Researchers have credited these advantages to the antioxidant characteristics of THCV and how the CB1 receptor blocks when the CB2 receptor is activated. The compound is said to also be able to help in areas of analgesic, anorectic, anti-emetic, anti-epileptic, anti-inflammatory, bone growth, and stimulant euphoriant.
We still don’t know enough about this compound, but it is one of the most promising cannabinoids.
Interesting Facts
- The chemical formula of THCV is C19H26O2.
- It has a mass of 286.4 g/mol.
- The IUPAC name of THCV is (6aR,10aR)-6,6,9-trimethyl-3-propyl-6a,7,8,10a-tetrahydrobenzo[c]chromen-1-ol.
References
PubChem [Internet]. Bethesda (MD): National Library of Medicine (US), National Center for Biotechnology Information; 2004-. PubChem Compound Summary for CID 93147, Tetrahydrocannabivarin https://pubchem.ncbi.nlm.nih.gov/compound/Tetrahydrocannabivarin
Husni, Afeef, “Optimization and Development of In Vitro Bioassays to Determine Structure-Activity Relationships for Cannabinoid Receptor 1 and Cannabinoid Receptor 2” (2012). Electronic Theses and Dissertations. 1459. https://egrove.olemiss.edu/etd/1459
Prandi C, Blangetti M, Namdar D, Koltai H. Structure-Activity Relationship of Cannabis Derived Compounds for the Treatment of Neuronal Activity-Related Diseases. Molecules. 2018 Jun 25;23(7):1526. doi: 10.3390/molecules23071526. PMID: 29941830; PMCID: PMC6099582.
Back to List of Cannabinoids